PRODUCT IDENTIFICATION
1071-93-8
TOXICITY
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
175 - 178 C
50 g/l
360 C
9.5 ((at 10g/l, 20 C)
GENERAL DESCRIPTION & APPLICATIONS
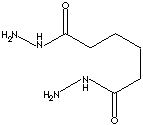
Hydrazine (anhydrous) is a clear, fuming, corrosive liquid with an ammonia-like odor; melting at 1.4 C, boiling at 113.5 C, specific gravity 1.011. It is very soluble in water and soluble in alcohol. It decomposes on heating or exposure to UV to form ammonia, hydrogen, and nitrogen, which may be explosive with a blue flame when catalysed by metal oxides and some metals such as platinum or Raney nickel. It is prepared from ammonia with chloramine in the presence of glue or gelatin (to inhibit decomposition of the hydrazine by unreacted oxidants) as the hydrate form usually (100% monohydrate contains 64% by weight hydrazine). Hydrazine is also prepared from sodium hypochlorite with urea in the presence of glue or gelatin. Botht ammonia and amines are nitrogen nucleophiles which donate electrons (they are Lewis bases). But hydrazine (diamine) has much stronger nucleophilicity which makes it more reactive than ammonia. Hydrazine has dibasic and very reactive properties. Hydrazine is used as a component in jet fuels because it produce a large amount of heat when burned. Hydrazine is used as rocket fuel. Hydrazine is used as an oxygen scavenger for water boiler feed and heating systems to prevent corrosion damage. Hydrazine is used as a reducing agent for the recovery of precious metals. It is used as a polymerization catalyst and a chain extender in urethane coatings. It and its derivatives are versatile intermediates. They have active applications in organic synthesis for agrochemicals, pharmaceuticals, photographic, heat stabilizers, polymerization catalysts, flame-retardants, blowing agents for plastics, explosives, and dyes. Recently, hydrazine is applied to LCD (liquid crystal displays) as the fuel to make faster thin-film transistors. Hydrazide is an acyl hydrazine. Acyl (-CO) is an organic radical formed by removal of a hydroxyl group from an organic acid (carboxyl group). Adipoyl hydrazide is used as polyurethane dispersion.
APPEARANCE
99.5% min (DRY)
95.0% min
0.1% max
MOISTURE
0.4% max
HYDRAZINE
500ppm max
Hazard Symbols: n/a, Risk Phrases: n/a, Safety Phrases: 24/25/28A/37/45